Organs are composites of tissue varieties with various developmental origins, and they depend on distinct stem and progenitor cells to fulfill physiological calls for for mobile manufacturing and homeostasis.
How various stem cell exercise is coordinated inside organs just isn’t effectively understood. Here we describe a lineage-restricted, self-renewing frequent skeletal progenitor (bone, cartilage, stromal progenitor; BCSP) remoted from limb bones and bone marrow tissue of fetal, neonatal, and grownup mice.
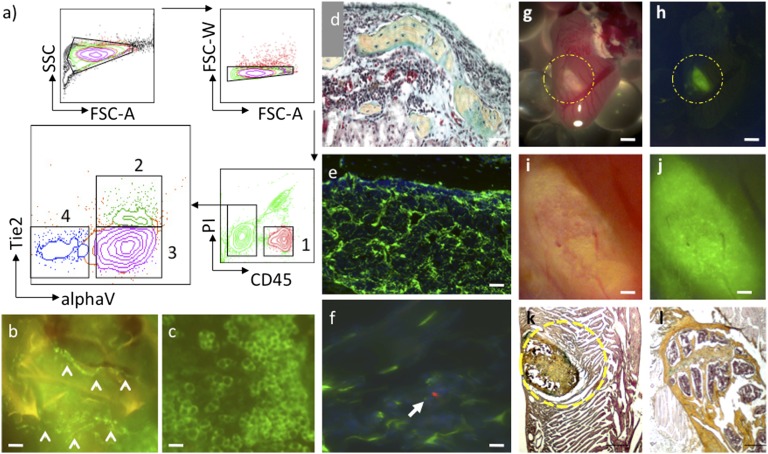
The BCSP clonally produces chondrocytes (cartilage-forming) and osteogenic (bone-forming) cells and a minimum of three subsets of stromal cells that exhibit differential expression of cell floor markers, together with CD105 (or endoglin), Thy1 [or CD90 (cluster of differentiation 90)], and 6C3 [ENPEP glutamyl aminopeptidase (aminopeptidase A)].
These three stromal subsets exhibit differential capacities to assist hematopoietic (blood-forming) stem and progenitor cells. Although the 6C3-expressing subset demonstrates purposeful stem cell niche exercise by sustaining primitive hematopoietic stem cell (HSC) renewal in vitro, the opposite stromal populations promote HSC differentiation to extra dedicated traces of hematopoiesis, such because the B-cell lineage.
Gene expression evaluation and microscopic research additional reveal a microenvironment through which CD105-, Thy1-, and 6C3-expressing marrow stroma collaborate to offer cytokine signaling to HSCs and extra dedicated hematopoietic progenitors.
As a outcome, inside the context of bone as a blood-forming organ, the BCSP performs a important function in supporting hematopoiesis by way of its technology of various osteogenic and hematopoietic-promoting stroma, together with HSC supportive 6C3(+) niche cells.
Hematopoietic stem cells (HSCs) are maintained in a selected bone marrow (BM) niche in cavities shaped by osteoclasts. Osteoclast-deficient mice are osteopetrotic and exhibit closed BM cavities. Osteoclast exercise is inversely correlated with hematopoietic exercise; nevertheless, how osteoclasts and the BM cavity probably regulate hematopoiesis just isn’t effectively understood.
To examine this query, we evaluated hematopoietic exercise in three osteopetrotic mouse fashions: op/op, c-Fos-deficient, and RANKL (receptor activator of nuclear issue kappa B ligand)-deficient mice.
We present that, though osteoclasts and, by consequence, BM cavities are absent in these animals, hematopoietic stem and progenitor cell (HSPC) mobilization after granulocyte colony-stimulating issue injection was comparable and even larger in all three traces in contrast with wild-type mice.
In distinction, osteoprotegerin-deficient mice, which have elevated numbers of osteoclasts, confirmed decreased HSPC mobilization. BM-deficient sufferers and mice reportedly keep hematopoiesis in extramedullary areas, comparable to spleen; nevertheless, splenectomized op/op mice didn’t present decreased HSPC mobilization.
Interestingly, we detected an HSC inhabitants in osteopetrotic bone of op/op mice, and pharmacological ablation of osteoclasts in wild-type mice didn’t inhibit, and even elevated, HSPC mobilization. These outcomes recommend that osteoclasts are dispensable for HSC mobilization and could perform as detrimental regulators within the hematopoietic system.
